COVID-19 — An Initial View
On WebChris Whitty at Gresham CollegeChris Whitty presents the then-current understanding of the 2019–2020 coronavirus pandemic. It covers its epidemiology and a few related historical epidemics. He presents a simple systematic approach to talking about viral epidemics, covering disease mortality, transmission and ends with possible treatments and vaccines.
COVID-19
The virus "SARS-CoV-2" originates from the Hubei province in China around the end of 2019. Its initial carriers were probably horseshoe bats, but since then it's become a human-to-human disease. It utilizes a common receptor in the cells of the human upper respiratory tract, ACE-2, to gain entry. It ends up causing COVID-19, a respiratory disease. For mild or early COVID-19, this includes the following symptoms.
- Fever
- Cough
- Myalgia (muscle aches)
- Headaches
- Shortness of breath
- Chills
- Sore throat
- Losing sense of smell or taste
Majority of symptomatic people have a fever or a cough. They don't need to seek medical assistance unless they deteriorate. They must, however, self-isolate. Most recover within 7 days, while the cough may remain for a few more weeks. More severe cases have respiratory failure, requiring oxygen. Organ failure is probably an immunological reaction and not directly caused by the virus. Critical cases have rapid deterioration. Risk of clots like pulmonary embolism and strokes. 25% of people requiring ventilation also require renal support due to kidney or multi-organ disease.
Spread
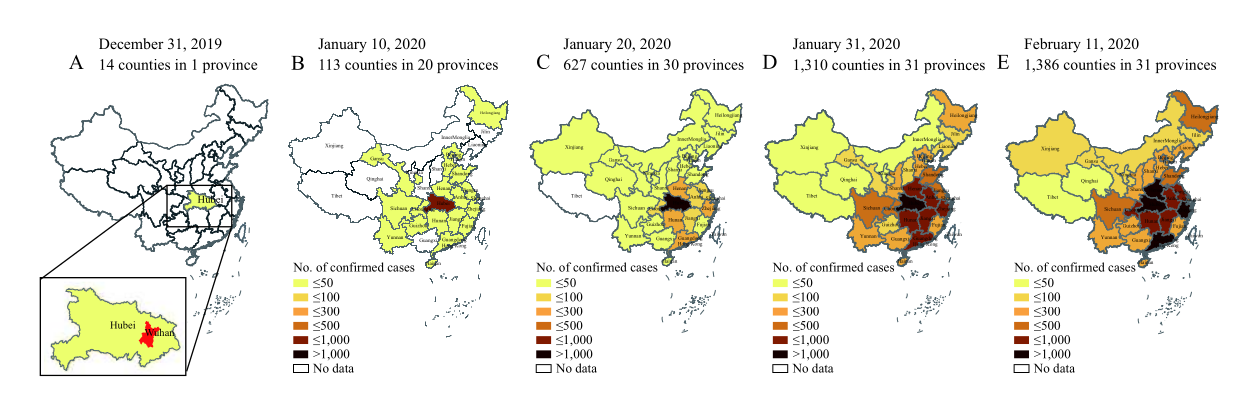
Its initial spread was very quick. In a matter of 43 days it moved from 14 counties to 1386 counties in China.
Spread of COVID-19 from Dec 31, 2019 to Feb 11, 2020. From "The Epidemiological Characteristics of an Outbreak of 2019 Novel Coronavirus Diseases (COVID-19) — China, 2020".
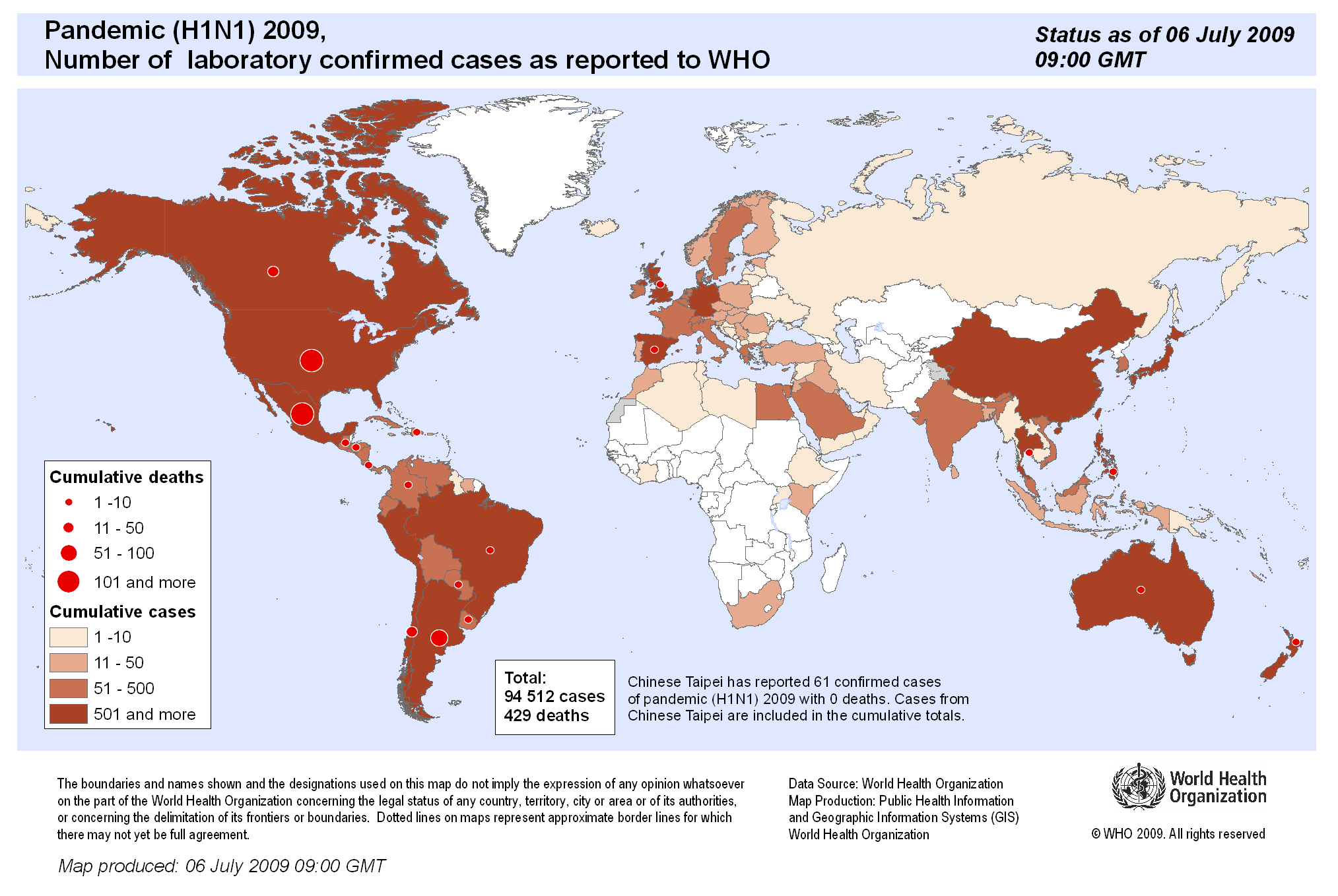
Initially few outbreaks beyond China, but by March 11 it was declared a pandemic by the World Health Organization. Today, as of May 7, 2020, its world spread looks like:
Testing
As the genome of the virus was found early, a direct virus test was rapidly developed utilizing the PCR method. This permits identifying infected people only while they're infected.
Alternatively there are serology tests, which identify antibodies. These help find out how many people have had the disease, even those lacking symptoms. It's unknown whether having SARS-CoV-2 antibodies implies immunity or lack of future infectiousness. There are some reports of people having been reinfected. The duration of possibly immunity is therefore unknown. There are multiple different antibodies, some of which are discoverable only 3–4 weeks after the disease.
For larger testing efforts the question of sampling strategies needs resolving, especially regarding asymptomatic cases. Faster tests are still needed to quickly identify people coming to hospitals.
Systematic Approach
Whitty presents a system approach to talking about epidemics:
- Mortality and severity (virulence)
- Route of transmission
- Force of transmission
- Treatments and vaccines
Out of the 5, we know we lack available treatments and vaccines for COVID-19.
Mortality
Mortality is determined by how many people get infected and of those, how many die (fatality rate). Infection fatality rate (IFR) for COVID-19 seems to be around or below 1%. This could be adjusted once we discover how many people are asymptomatic, infected without knowing it. Compared to pandemics below, it's fairly low:
| Disease | Infection fatality rate |
|---|---|
| Ebola | 70% |
| HIV | 100% when first emerged |
| Smallpox | 30% |
| Swine flu from 2009 (H1N1) | 0.1% |
| Spanish flu from 1918 (H1N1) | ~3% |
However, even a disease with low mortality, if it infects large numbers of people, ends up killing more people than diseases with higher fatality rates.
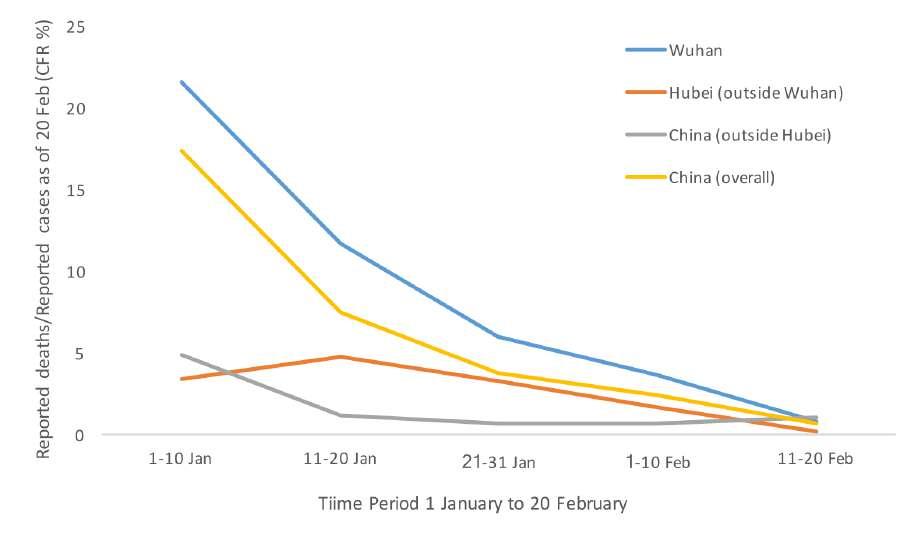
Initially, COVID-19s case fatality rate (chance of dying if you do develop symptoms as opposed to merely being infected) was high. That's often to be expected, as with new diseases people tend to notice deaths and severe cases. Only later will milder cases be included. As can be seen from the graph below, the assumed mortality changed over time as more cases were identified.
Assumed mortality from World Health Organization joint mission to China report
Other Coronaviruses
Up to 15% of ordinary colds and the occasional pneumonia are caused by 4 other coronaviruses:
- 229E (alpha coronavirus)
- NL63 (alpha coronavirus)
- OC43 (beta coronavirus)
- HKU1 (beta coronavirus)
Over the years, two others have arisen:
Risk of Death
Individually the chance of dying from COVID-19 are low.
- A high proportion of people will not get it.
- A significant proportion of people will have no symptoms.
- Great majority (~80%) of symptomatic cases develop to a mild to moderate disease.
- A minority need hospitalization, mostly only oxygen. Majority will survive.
- Minority of the prior will need ventilation.
- Minority of every age-group die, however even in a risk-group majority do not.
Risk factors could be categories into three — age, gender and comorbidities.
Age
The biggest risk factor is age. The case fatality rate (chance of dying if you develop symptoms) of all age groups is below.
| Age group | Case fatality rate |
|---|---|
| 0-9 | 0.003% |
| 10-19 | 0.01% |
| 20-29 | 0.06% |
| 30-39 | 0.15% |
| 40-49 | 0.3% |
| 50-59 | 1.25% |
| 60-69 | 4% |
| 70-79 | 8.6% |
| 80+ | 13.4% (IFR 7%?) |
From "Estimates of the severity of coronavirus disease 2019: a model-based analysis", with replication data on GitHub and supplementary appendix.
The numbers above seem to match data from other countries, such as England and Wales, Italy and Spain. You see very few deaths among those below 50.
Keep in mind that the infection fatality rate, chance of dying if you're infected, could differ from case fatality rate, the chance of dying if you develop symptoms. The ratio for COVID-19 is still unknown.
Older people are generally far more likely to die of infection than other age groups, although that doesn't hold universally. The 1918 Spanish flu pandemic hit the very young in addition to the very old. Middle aged adults (ages 24–34) lied somewhere in between.
While COVID-19 does increase the chance of dying in any age group, the baseline for the very young is extremely low to begin with, thereby minimally increasing their overall risk. That cannot be said for older people, who bear the highest risk of getting the severe form of COVID-19 and are therefore also more likely to die.
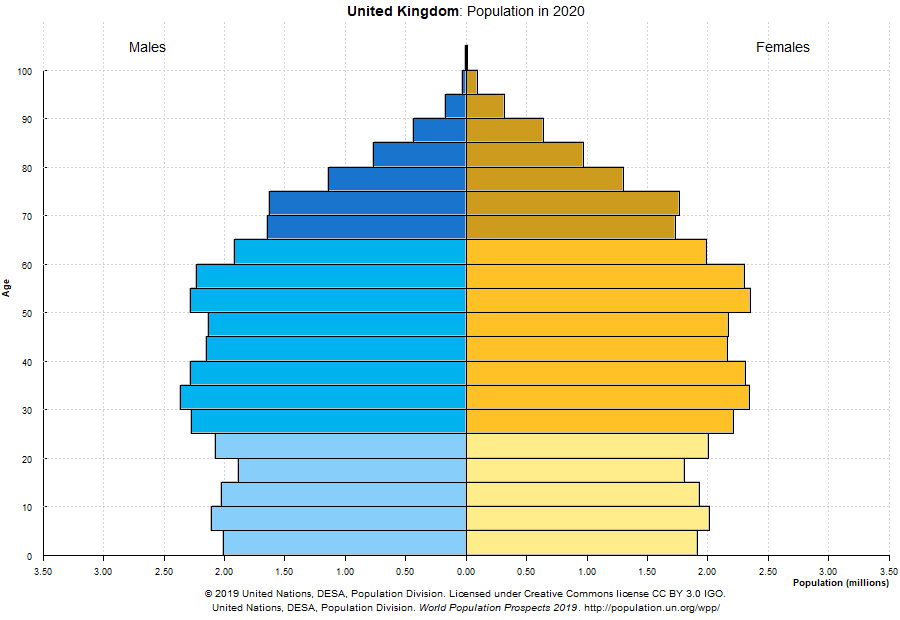
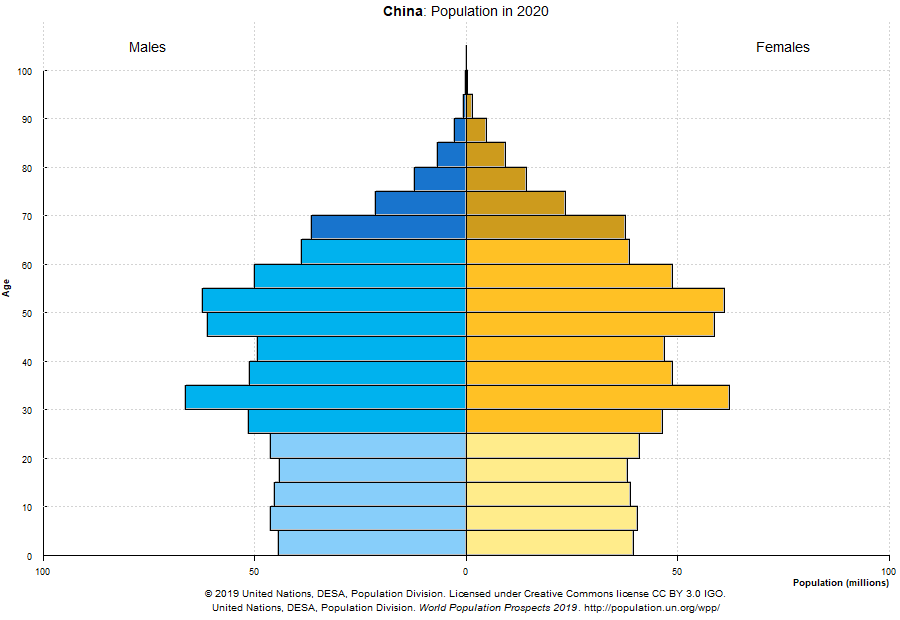
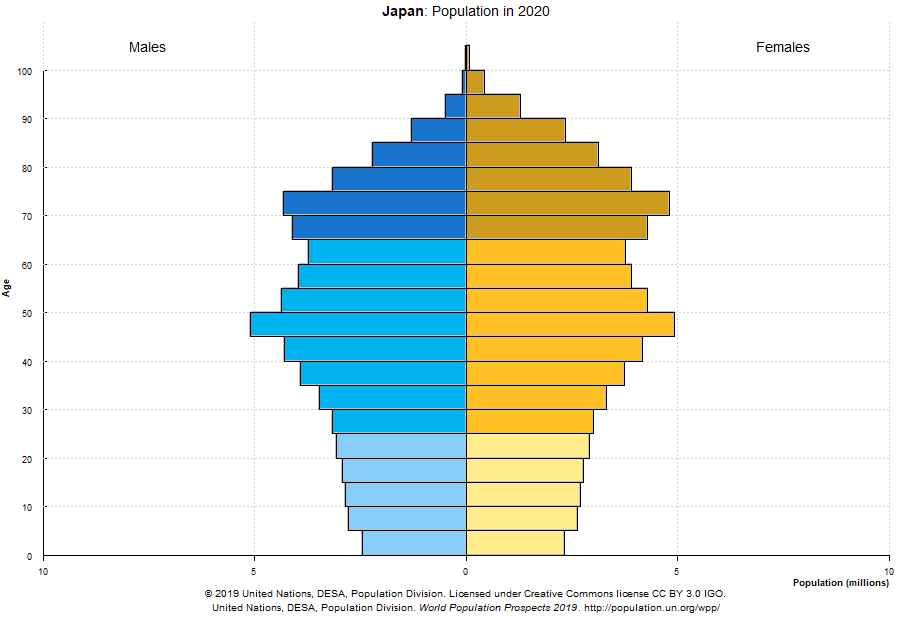
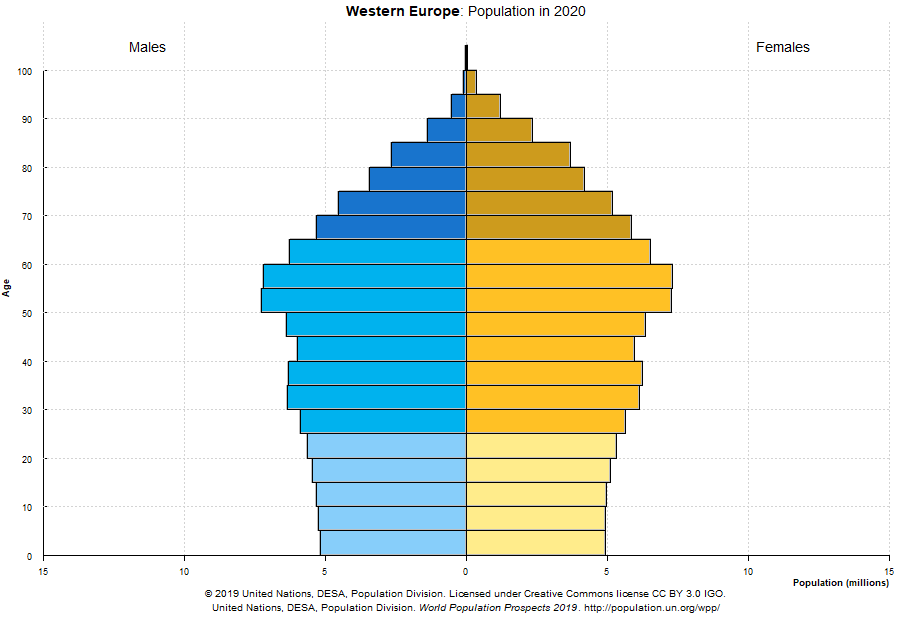
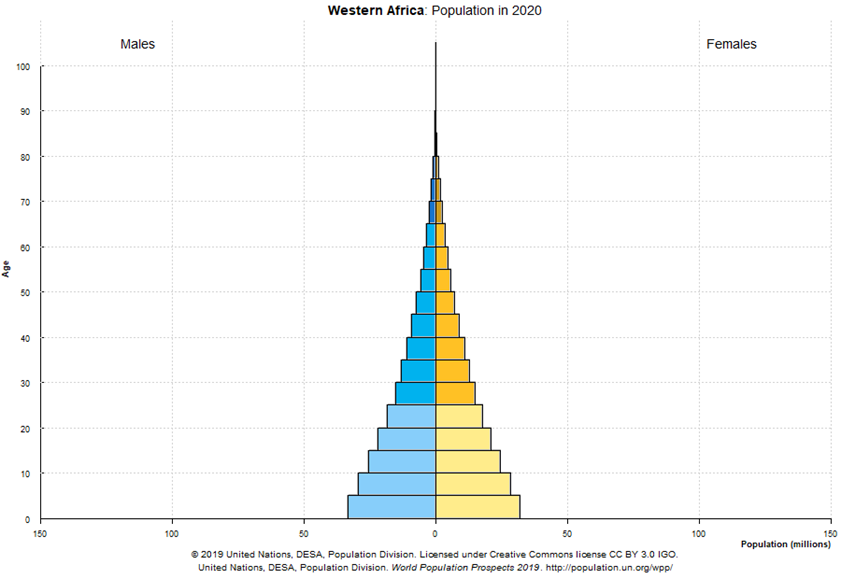
Demographics play an important role in determining how prevalent severe cases are in any given country. The following distributions could help explain why some countries were or will be hit more severely, even if the rate of infections (percentage of total population infected) is comparable.
Gender
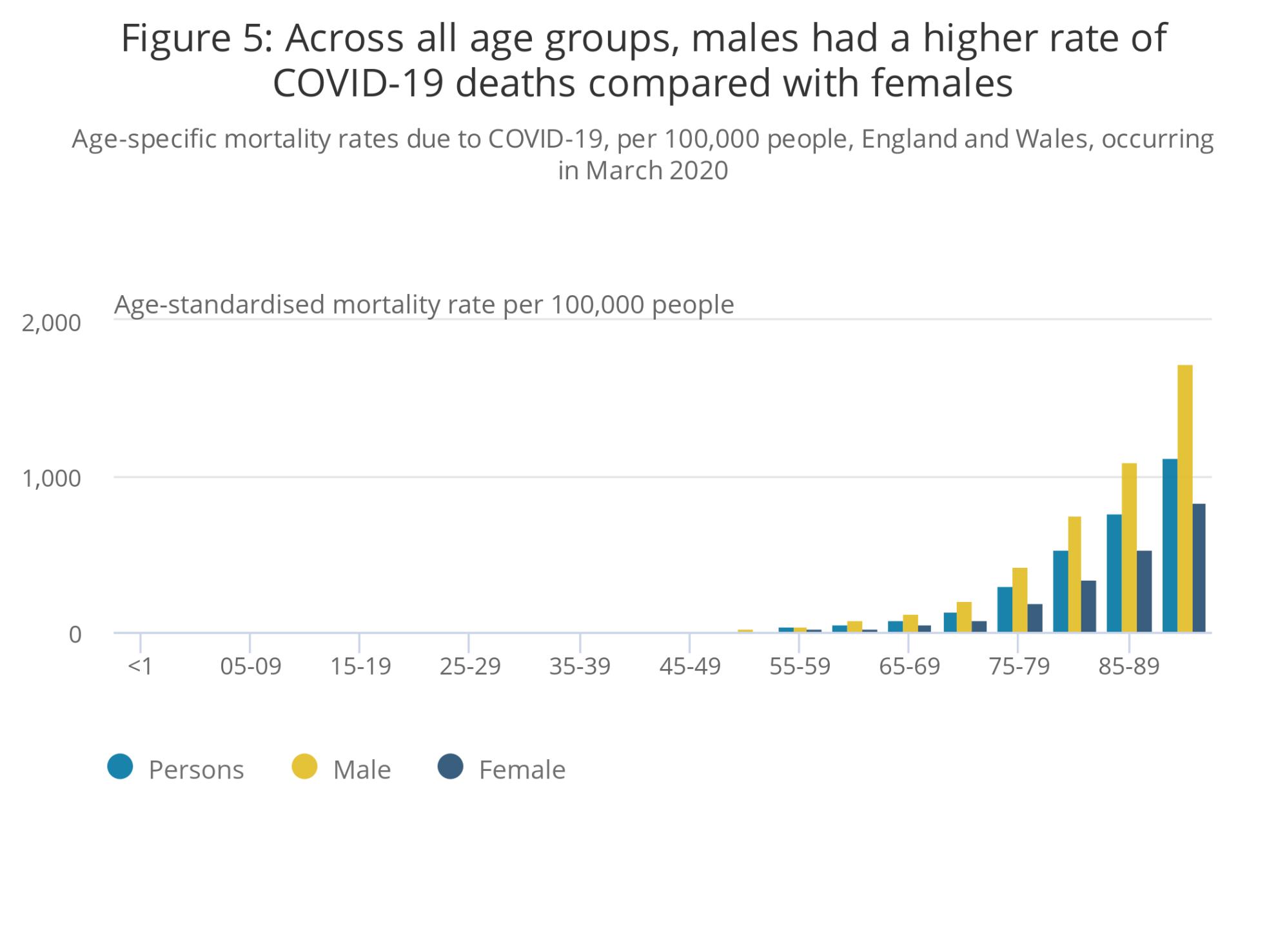
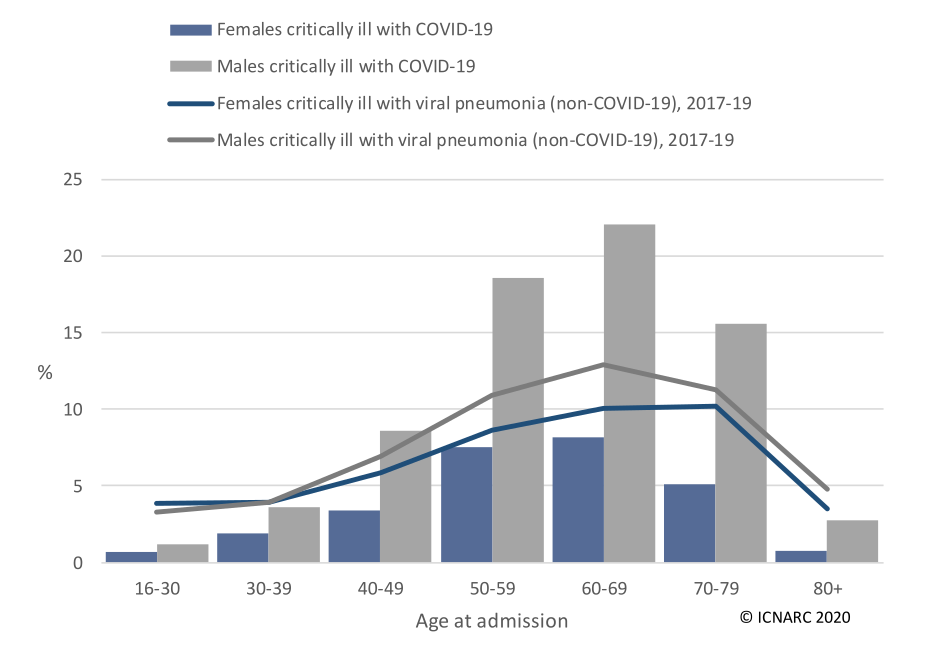
Somewhat surprisingly, COVID-19 affects men more than women, something that's not the case with other viral pneumonias.
Data from the Office for National Statistics "Deaths involving COVID-19, England and Wales: March 2020" dataset.
The gender difference is especially pronounced when looking at patients requiring intensive care.
Comorbidities
Comorbidities are other diseases that make people more susceptible to COVID-19. In the UK's Office for National Statistics data, 91% of cases had at least one of the following comorbidities:
- Cardiovascular disease
- Diabetes
- Chronic lung disease
- Immunosuppression
- Chronic kidney and liver disease
- Obesity
In addition, some ethnic groups may be more at risk, either due to some comorbidities being more likely within particular groups, their socio-economic status or even genetics.
Indirect Deaths
In addition to direct deaths from the disease given a functional health-care system and optimal care, there are 3 other ways how a pandemic could cause mortalities.
- Indirect deaths due to health system getting overwhelmed and unable to treat both COVID-19 and other disease patients.
Hasn't occurred in the UK. - Indirect deaths through postponed treatment, screening or elective surgeryelective-surgery or general fear of going to the hospital.
- Interventions put in place which have a strong economic impact.
Increased deprivation and ill health are strongly correlated.
Low-Income Countries
For most infections, poverty and malnutrition are correlated with worse outcomes. It's possible mortality rate will be higher in middle and lower income countries. Weaker health systems are more easily overwhelmed. However, occasionally middle and lower income countries have better public health care than expected.
Route of Transmission
Primary routes of possible transmissions:
| Method | Example disease |
|---|---|
| Respiratory | COVID-19, SARS, MERS, flu |
| Sexual or blood | HIV |
| Touch | Ebola |
| Water or food | Cholera |
| Vector (insects, parasites) | Zika, malaria |
Respiratory viruses are most difficult to tackle. They mainly enter via the nose, mouth or possibly eyes. Droplets above 5–10µm come from coughing, sneezing or talking. They're usually carried no more than a meter. Additionally, SARS-CoV-2 could be transmitted via a secondary route by touching contaminated surfaces and then your face.
Ways to reduce respiratory transmission would be to catch your cough and washing your hands with soap or alcohol gel. Soap damages the lipid around the virus. The majority of SARS-CoV-2 will have been gone by 72h on various surfaces, with porous surfaces like cardboard reducing while flat surfaces like plastics or stainless steel prolonging its lifespan.
Some viruses are infectious only when symptoms develop. Unfortunately SARS-CoV-2 seems to be infectious even 2–3 days prior to getting symptoms and infectious even if its asymptomatic. Once people do develop symptoms, it probably takes about a week before the infectiousness drops off significantly. This was investigated in "Temporal dynamics in viral shedding and transmissibility of COVID-19". Infectiousness even in asymptomatic cases means merely isolating those visibly ill won't be sufficient.
SARS
The severe acute respiratory syndrome has similarities to today's strain, with origins in bats around 2002–2003. Around 8422 were infected at that time, out of which 11% died. Largest outbreak, 63% of total cases, was in China. 20% in Hong Kong. Low hundreds in Taiwan, Canada and Singapore. Very few cases elsewhere. Transmission was probably exclusively from people who had symptoms. Hasn't returned since.
MERS
The Middle East respiratory syndrome jumped from dromedaries, Arabian camels, to humans around 2012. It too originates from bats, even if camels were the intermediate step. Had a high — 35% — death rate. Had one outbreak in South Korea in 2015 (185 confirmed cases), but didn't take off beyond that.
Spanish flu
From 1918–1919, the H1N1 influenza virus caused the death of 17 million up to 100 million people. It did so in 3 waves over 2 years, with the second wave being more deadly than the first.
H1N1 is of the same family that's responsible for the 2009's swine flu, albeit a new strain. The latter strain caused around 43–89 million cases and 8–18 thousand deaths. High transmission, but low mortality.
Asian flu
H2N2 resulted in the 1957 Asian Flu pandemic, which had a ~0.6% mortality and killed around 1 to 4 million people worldwide.
Force of Transmission
R0 or force of transmission is how many people one infected person in turn infects. For an epidemic and exponential growth to occur, R0 has to be above 1. Here's how COVID-19's R0 in the United Kingdom stacks up against other diseases:
| Disease | R0 |
|---|---|
| COVID-19 | 3 |
| Ebola | 1.2–2.5 |
| Spanish flu from 1918 | 2–3 |
| HIV | 2–5 in the expansion phase |
| Measles | >10 |
| Malaria | >100 |
Reducing Transmission
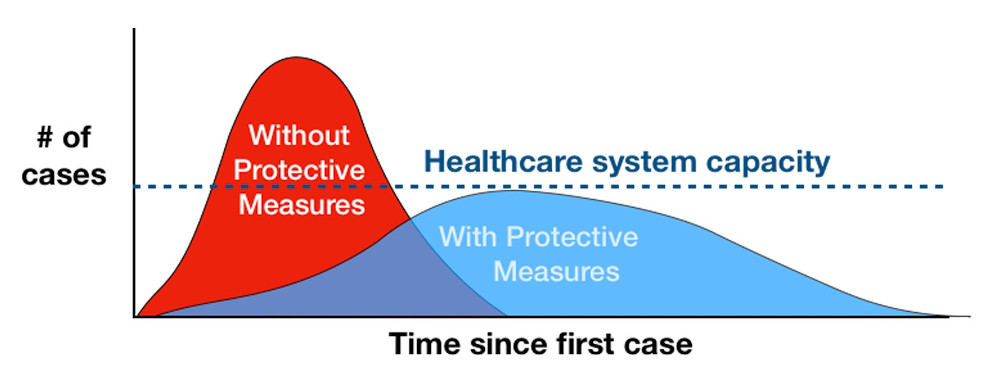
In the absence of a vaccine or drug, relying on social and public health measures could help address the quick growth. One popular theory is "flattening the curve", which is an attempt to reduce the transmission rate through social measures for an extended period of time in hopes of giving the heath-care system time to handle existing cases and possibly expand its capacity.
Without vaccines or drugs, there are four approaches:
- Reduce chance of infection through hygiene.
- Isolate cases and close contacts or family.
- Reduce contact between households via social distancing.
- Reduce chance of infection of the most vulnerable.
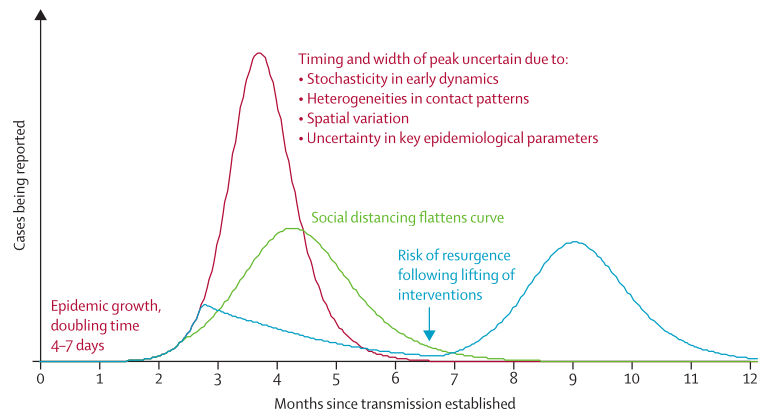
Its apparently shown that a combination of the measures above are necessary, as none individually are sufficient. There is, however, a risk of R0 returning to levels above 1 once the measures are lifted. There's a possibility those measures will have to be continued indefinitely until medical countermeasures have been developed or there's evidence a large number of people have been infected asymptomatically.
From "How will country-based mitigation measures influence the course of the COVID-19 epidemic?".
The optimal answer on how and when to unwind lockdowns is dependent on whether to focus on direct or indirect deaths. There are important social and economic considerations. Alluding to the flu pandemics earlier, it's possible for the second wave to be more dangerous than the first. That's why it's important to ensure R0 does not get above 1.
Seasonality
There may be a seasonal element to COVID-19. While winter has always put health-care systems under more pressure due to higher transmissions of respiratory viruses, the current pandemic may exaggerate that. On the other hand, social distancing measures already in place may reduce flu and other upper respiratory tract infections. Observing or identifying COVID based on symptoms may get harder during the winter season as there are plenty of seasonal diseases with similar symptoms. Overall, though, the climate's (temperature, humidity) impact is unknown.
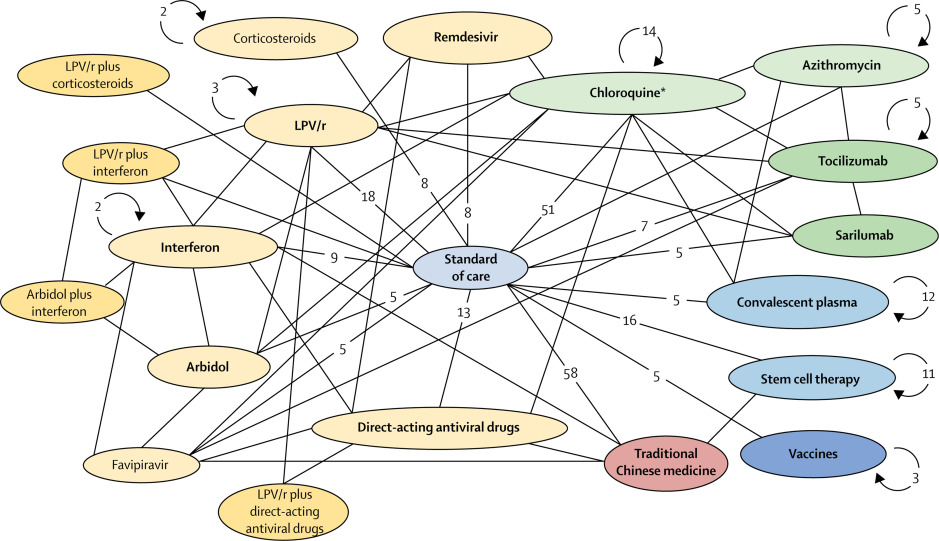
Treatments and Vaccines
Vaccines
Vaccine strategies come in multiple different forms:
-
Epidemic modifying vaccines are highly effective at preventing infection. Giving it to the majority of the population means even the few that weren't vaccinated are unlikely to be infected as people around them won't pass the virus on.
-
Disease modifying vaccines may be only partially effective. They're most useful for high risk groups, as long as they reduce the severity of the disease and complications.
Drugs
Many diseases lack vaccines, but can be adequately dealt with by drugs. This could help people with existing mild symptoms from becoming severely ill. Alternatively only severe cases could be treated, avoiding need for intensive care and possible death.
Possible treatments can be grouped into three categories:
-
Antivirals
Useful early or as prophylaxis. Generally less well developed than antibiotics, although we do have moderately good antivirals for influenza, decent antivirals to suppress HIV or even cure Hepatitis C. -
Anti-inflammatory drugs or immune modulating drugs
There are many feedback loops in the immune system. We've got powerful drugs that can suppress some parts of it, such as steroids, drugs from rheumatology and drugs for organ transplants.Severe COVID-19 could be from cytokine storm, which is a runaway effect of white blood activating more white blood cells, which then end up causing more damage to the body than the virus itself. If it truly is an auto-immune disease, anti-inflammatory drugs could help. It's possible they could make things worse though.
-
Antibodies
Take the antibodies from recovered people and inject them to patients. This presumes recovered people have antibodies. Eventually we may be able to manufacture antibodies.
While it may seem appealing to begin high-dose treatment given a plausibly effective drug, it's important to do trials before doing so. One study, "Effect of High vs Low Doses of Chloroquine Diphosphate as Adjunctive Therapy for Patients Hospitalized With Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection", apparently found that high doses of chloroquine actually caused more deaths than low doses.
Unknowns
- Proportion of asymptomatic infections.
- Immunity duration after recovery
- Blood test correlation with immunity
- Children's transmission rate. Relevant for opening schools.
- Seasonality
- Pathological reason for deterioration after a week.
- Gender differences in severe cases.
- Elective surgery being non-emergency surgeries, but still done to preserve health.↩